Right Patient. Right Time. Right Line.
Vascular Access requires timely assessment, planning, insertion and follow-up. Appropriate device selection can impact the length of stay and complications, therefore, early assessment of a patient who requires a vascular access device should be considered upon admission.
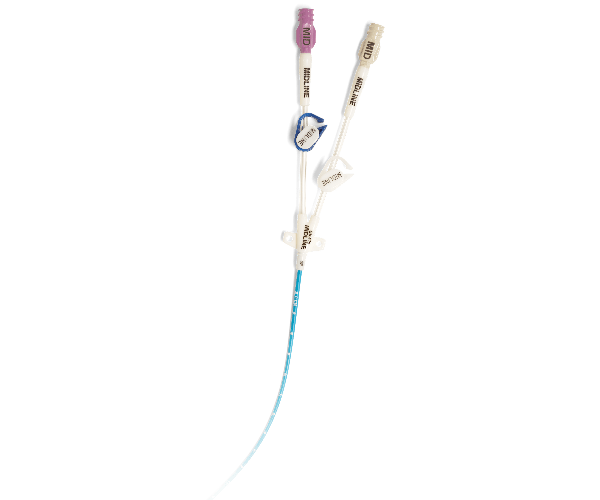
The BioFlo Midline Catheter is an effective solution to preserving a patient’s peripheral access. It provides a cost-effective alternative to multiple IV site rotations for patients who need short-term venous access. This improves the likelihood that the patient will receive his or her ordered therapy, which may lead to a shorter hospital stay, and improved patient satisfaction.
Proven to Reduce Thrombus Accumulation, In Vitro
The BioFlo Midline Catheter is the only midline catheter with Endexo Technology, providing a catheter material more resistant to platelet aggregation1. This novel, disruptive technology is present throughout the catheter shaft including the intraluminal, extraluminal and cut surface of the catheter, and is present for the life of the catheter.
BioFlo Midline Catheter
- X-ray confirmation is not required after placement
- Visibly marked as a midline catheter to aid in identification
- Multiple kit configurations to meet clinician needs:
- Maximal Barrier Nursing Kit
- MST KitCatheter Only Kit
- Available with standard stylet or nitinol guidewires only
- Power Injectable