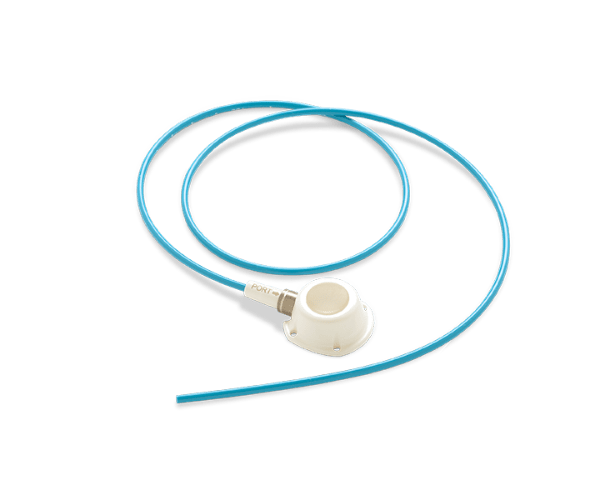
Proven to Minimize Thrombus Accumulation.
Make protection part of the mix.
Endexo Technology is a permanent and non-eluting polymer that is “blended” into the polyurethane from which the catheter is made. It is present throughout the catheter including the extraluminal, intraluminal and cut catheter surface of the tip. Endexo Technology remains present for the life of the catheter and provides a catheter material more resistant to thrombus accumulation.
ADDITIONAL FEATURES:
- CT-rated up to 5mL/sec and 300 psi and MRI-conditional—3 Tesla.
- Radiopaque under fluoroscopy
- Flexible, kink-resistant catheter material
- 1/2” septum diameter for ease of port access
Available with PASV Valve Technology, designed to resist backflow, maintain long-term patency, and provide a saline-only flushing option.
EASILY IDENTIFY CRITICAL INFORMATION
- Radiopaque “CT” lettering confirms if port is power injectable or flipped
- Patient reminder band, ID card, key tag and record sticker provide added verification that the port is power injectable